6) To complete the sandwich, one more piece of paper goes on and then the last pad, again making sure both are thoroughly soaked in TB. When it is completely soaked with TB, move it onto the sandwich next. 5) Use the tweezers to grab the nitrocellulose membrane and dunk it in the dH2O container, make sure the entire membrane is wet, and then move it to the TB. Then put on a pad and one piece of paper (make sure both are completely wet in the TB). 
4) To make the blotting sandwich, begin by putting the blotting apparatus in the TB container with the black side down. To remove the other plate, place the gel in the TB container and gently scrap off with a flat scoopula. 3) Remove one glass plate from the gel by twisting a spacer between the plates. Set up blot: 2) Prepare two tupperware containers, one with dH2O and one with 1/2X Transfer Buffer (TB). Cover the membranes with chemiluminescent substrate ( GlObuffer ), acquire images using CCD cool ed camera.Western Blotting 1) Run protein gel.I ncubate with appropriate secondary antibody diluted in 1X DiluObuffer (FabGennix Secondary antibody conjugates are diluted 1:15,000-25,000 in DiluOBuffer for optimal signal to noise ratios on blots).After 1 hour, wash 3X with 1X TBST (10 minutes each time).If the antibody was diluted in DiluObuffer, the remaining antibody can be stored at 4oC for reuse. The casein/BSA and other proteins will bind Igs and reduced the effective concentration of antibody for blotting. Note: If the antibody dilution buffer contains milk/BSA or ovalbumin, increase the concentration of primary antibody.Incubate the membranes with primary antibodies for 1 hour. Dilute the primary antibodies at 1:250-1:1000 in 3ml of antibody dilution buffer (1X DiluObuffer ) per 1 cm 2 of membranes.Wash one more time in 1XPBS for 5 minutes on shaker. 
After blocking, rinse the membrane twice in 1XTBST and wash twice for 5 minutes each time in 1X TBST on shaker.Note: If blocking buffer contains Casein then it must be washed with PBS or TBS, this step will increase the signal to noise ratios on the blot.Place the membrane in blocking buffer (5X DiluObuffer ) for 1 hour on shaker at room temperature or overnight at 4 o C.Stain with Ponceaus to assess the quality of transfer and to mark the MW markers on membrane.For high MW/large glycosylated proteins, reduced the concentration of methanol form 20% to up to 15% and transfer for longer times (plate electrodes: Limit 250mA at 50 volts for 7 hours) the conditions are different for wire electrodes, overnight transfers will be preferred for HMW proteins at low methanol concentrations). 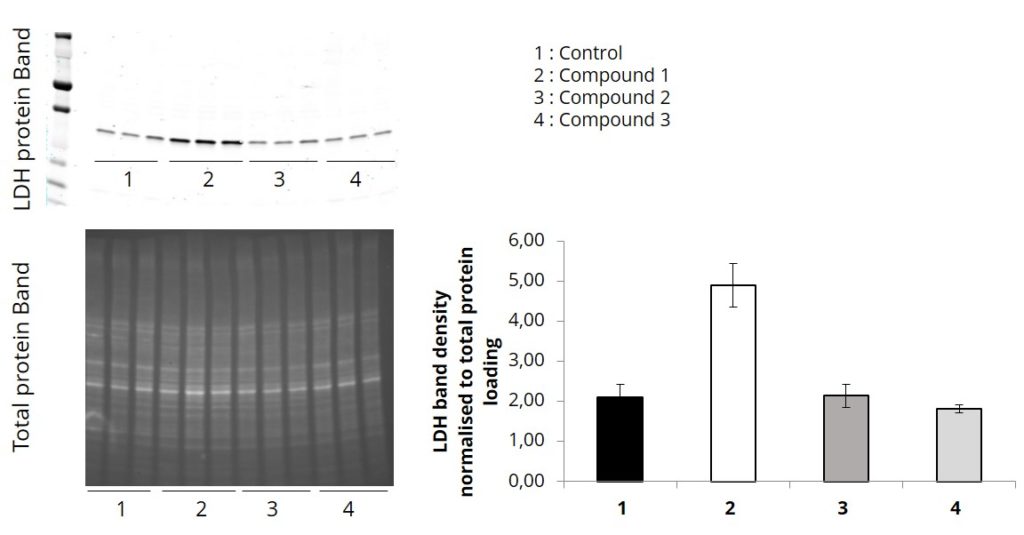
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
